James Chambers has compiled a collection of papers examining health benefits (and cost-effectiveness) using the Tufts Cost-Effectiveness Analysis Registry. Across these studies, he found that treatments for rare diseases, specialty drugs, and drugs that received expedited review were more likely to provide the greatest health benefit (gaining ≥0.9 QALYs compared with control drugs).
Chambers et al. (2020) The same is shown for treatments for rare diseases:
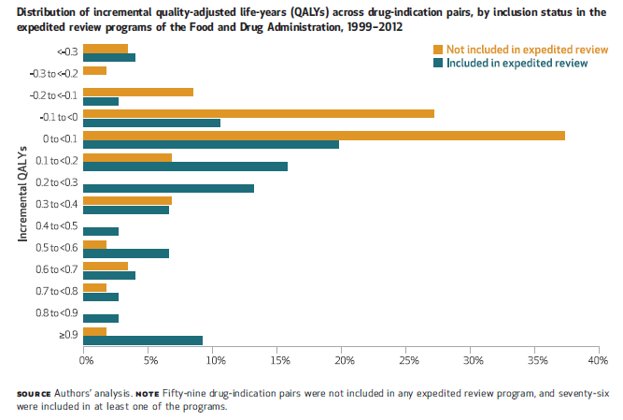
Chambers et al. (2017) This was shown to be true for treatments subject to expedited review:
and Chambers et al. (2014) The same is shown to be true for specialty drugs.
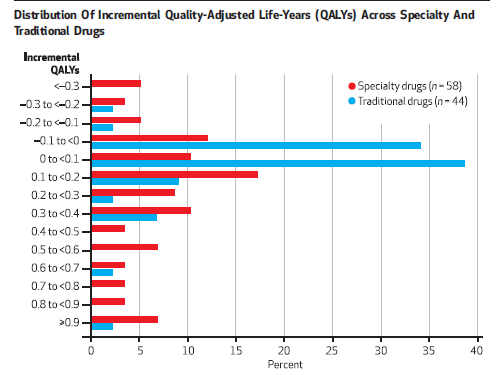
Note that greater health gains are often associated with higher drug prices. For rare diseases, these highly beneficial treatments are unlikely to be cost-effective due to the high cost of treatment. This is not surprising, however, as rare diseases by definition have smaller markets and therefore require higher prices to fund R&D efforts.on the other hand Chambers et al. (2014) Research shows that although specialty drugs cost more, the cost-effectiveness is comparable to conventional drugs because the average health benefits are higher.



